36 lewis diagram for nh3
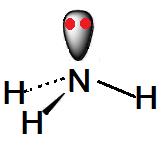
Ammonia 3d Structure. Here are a number of highest rated Ammonia 3d Structure pictures on internet. We identified it from well-behaved source. Its submitted by supervision in the best field. We tolerate this kind of Ammonia 3d Structure graphic could possibly be the most trending subject bearing in mind we portion it in google pro or facebook. Lewis Dot Diagram Of Nh3 The Lewis structure of ammonia, NH3, would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons. Electron Dot Structure of NH3 by Jeff Bradbury - February 17, - Lewis Electron Dot Structure for ammonia molecule NH3. by crator-avatar Jeff Bradbury 2.
Draw the Lewis Structure. Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons. … Name the electron-group geometry. … Looking at the positions of other atomic nuclei around the central determine the molecular geometry. What is the shape of ICl for negative?
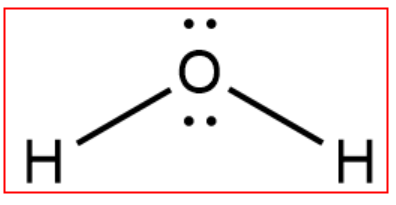
Lewis diagram for nh3
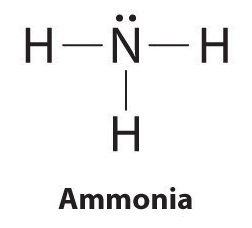
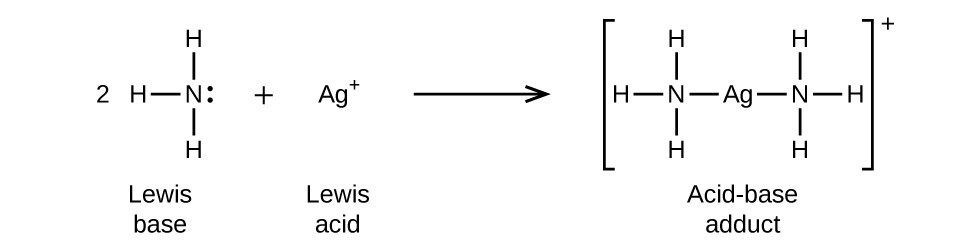
Draw the lewis diagram as below: Geometrical Structure of the Ammonia (NH3) The bond angle among the hydrogen-nitrogen-hydrogen atoms (H-N-H) is 107°. It is clear to understand that the geometrical structure of NH3 will be bent. Explanation: The Lewis structure of ammonia, NH3 , would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the atom. This is the reason why ammonia acts as a Lewis base, as it can donate those electrons. Likewise, what is the shape of nh3? trigonal pyramidal In this structure, as the name suggests, electrons are shown as dots and a line for bonding electrons between the two atoms. Let us now draw the Lewis dot ...
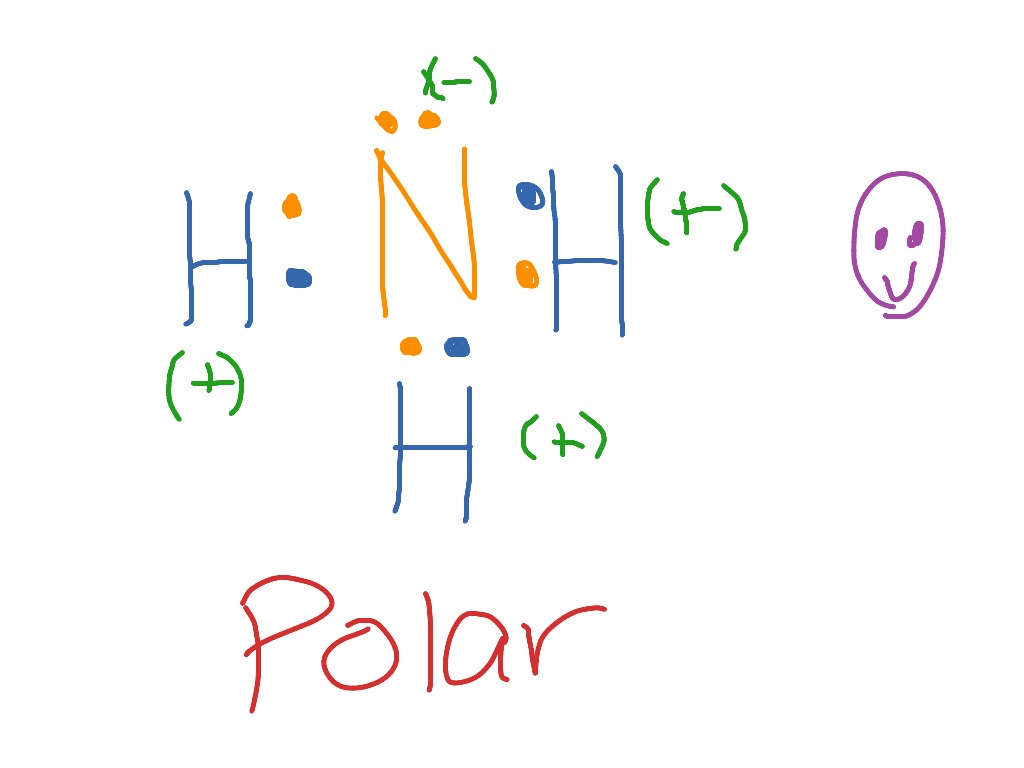
Lewis diagram for nh3. What intermolecular forces are present in NH3? You know that, ammonia is a polar molecules. it exhibits, dipole-dipole intraction, induced attraction, and London dispersion forces.NH3 is called dipole dipole because nh3 make N-H bond, it directly make hydrogen bonding. Ammonia (NH3) is a commonly tested Lewis structure due to it's widespread use in agriculture as a fertilizer. It also is a good example of a molecule with a ...25 Oct 2016 · Uploaded by Wayne Breslyn The structure looks like this: · First Nitrogen tends to form 3 covalent bond with 3 Hydrogen atoms which produces NH3. · After the formation of NH3 there is ...3 answers · 2 votes: Electrons of N is shared by H to complete its duplet and elctrons of H is shared by N to ... Aggregate Diagram. Here are a number of highest rated Aggregate Diagram pictures on internet. We identified it from obedient source. Its submitted by meting out in the best field. We assume this kind of Aggregate Diagram graphic could possibly be the most trending topic subsequent to we allowance it in google improvement or facebook.
Ammonia lewis structure contains three N-H bonds and one lone pair on nitrogen atom. Lewis structure of NH3 can be drawn by using valence electrons of ... In the lewis structure of ammonia (NH 3), there are three N-H bonds and one lone pair on nitrogen atom. Lewis structure of NH 3 can be drawn by starting from valence electrons of nitrogen and hydrogen atoms in several steps. Each step of drawing the lewis structure of NH 3 is explained in detail in this tutorial. Ammonia 3d Structure. Here are a number of highest rated Ammonia 3d Structure pictures upon internet. We identified it from reliable source. Its submitted by organization in the best field. We receive this kind of Ammonia 3d Structure graphic could possibly be the most trending topic in the manner of we ration it in google plus or facebook. The Lewis structure for NH3 is.The Lewis structure of ammonia, #NH_3#, would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the schematron.org is the reason why ammonia acts as a Lewis base, as it can donate those electrons.
Lewis structure of NH3 can be drawn by starting from valence electrons of nitrogen and hydrogen atoms in several steps. Can you draw the electron dot structure of ammonia? Out of which 3 electrons of nitrogen form a covalent bond with hydrogen, with sharing of one electron of nitrogen and one electron of hydrogen. There are three such bonds. 5 May 2018 — The Lewis structure of ammonia, NH3 , would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons ...1 answer · Have a look here... Explanation: The Lewis structure of ammonia, NH3, would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone ... A step-by-step explanation of how to draw the NH3 Lewis Dot Structure (Ammonia).For the NH3 structure use the periodic table to find the total number of vale... Unit 1 lecture 1 Introduction speaker: Wang Yifan,Yan Jun 1 Definition and main branches of chemistry随堂测验 1、1. Among the main branches of chemistry, the so-called "four major chemistry" secondary disciplines are
NH3 Lewis Structure Molecular Geometry. Gallery of Ammonia 3d Structure. St Augustine Homes For Sale Best Automatic Pool Cleaner Cat Asthma Treatment Purina Large Breed Puppy Food Honda Ridgeline 2008 Hotels Pueblo Co Can Dogs Eat Turkey Bacon Cat That Looks Like A Cheetah Wags And Wiggles Delta Dental Arkansas Diesel Mechanic Jobs Near Me ...
This chemistry video tutorial explains how to draw the lewis structure of NH3 also known as Ammonia.My Website: https://www.video-tutor.netPatreon: https:/...
In this model of a molecule of ammonia NH3 how many covalent bonds are represented. Bonds created by sharing electrons with other atoms. Hannahsonnentag Weebly Com Covalent bond diatomic molecule Lewis diagram molecule noble gases nonmetal octet rule shell valence valence electron Prior Knowledge Questions Do these BEFORE using the Gizmo Note.Covalent bonds gizmo quiz …
What is the shape of BrF 2? Bromine atom has seven valence electrons. Out of these, five from shared pairs with the electrons of fluorine atoms while the remaining two electrons constitute one lone pair. In order to minimise, the repulsive interactions in these electron pairs, the shape of the molecule is distorted octahedral or […]
In this structure, as the name suggests, electrons are shown as dots and a line for bonding electrons between the two atoms. Let us now draw the Lewis dot ...
Explanation: The Lewis structure of ammonia, NH3 , would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the atom. This is the reason why ammonia acts as a Lewis base, as it can donate those electrons. Likewise, what is the shape of nh3? trigonal pyramidal
Draw the lewis diagram as below: Geometrical Structure of the Ammonia (NH3) The bond angle among the hydrogen-nitrogen-hydrogen atoms (H-N-H) is 107°. It is clear to understand that the geometrical structure of NH3 will be bent.








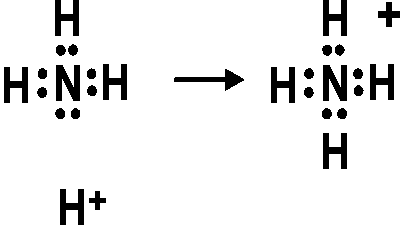




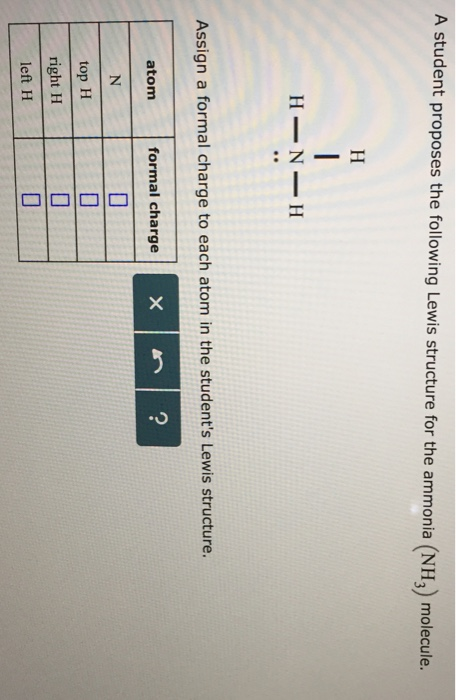




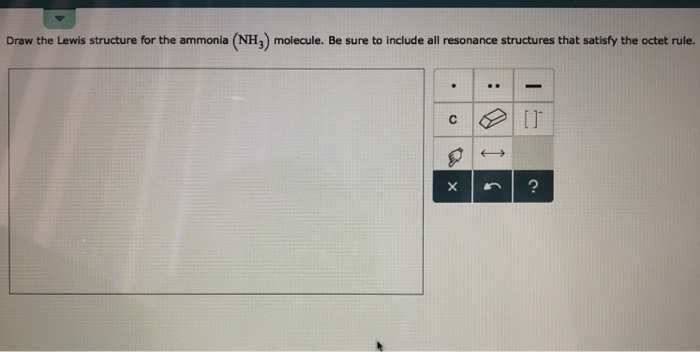







0 Response to "36 lewis diagram for nh3"
Post a Comment