41 c2- molecular orbital diagram
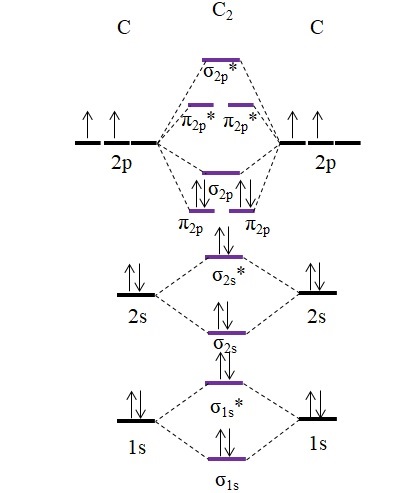
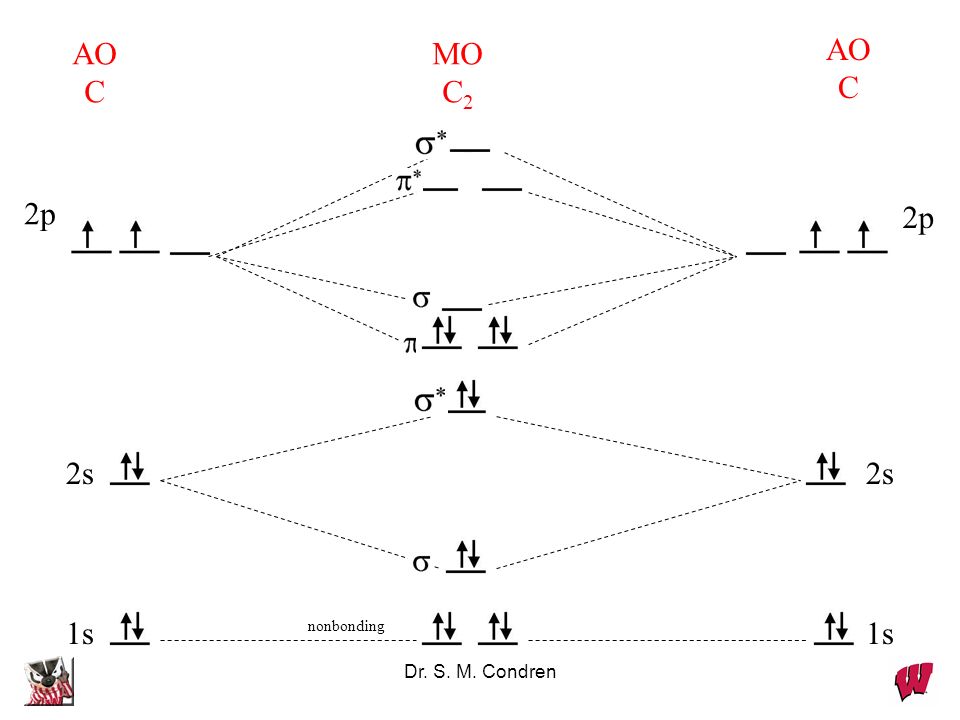
C2 molecular orbital diagram. A diatomic molecular orbital diagram is used to understand the bonding of a diatomic molecule. Get 11 help now from expert chemistry tutors. Fill from the bottom up with 8 electrons total. They also give insight to the bond order of the molecule how many bonds are shared between the two atoms. 26 Feb 2018 — write molecular orbital configuration of c2+ predict magnetic behaviour and calculate its bond order. ... The C2 molecule is diamagnetic because ...
When two carbons atoms bond, the pi(2p) bonding molecular orbitals are lower in energy than the sigma(2p) bonding orbitals.C2(2-) has a bond order of 3, so i...
C2- molecular orbital diagram
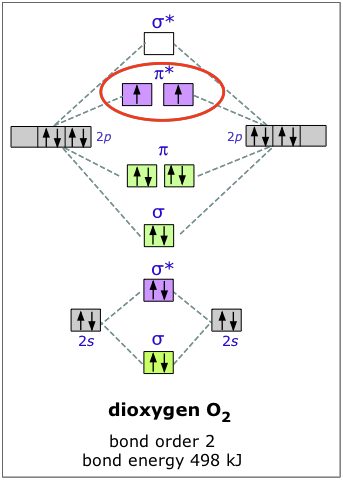
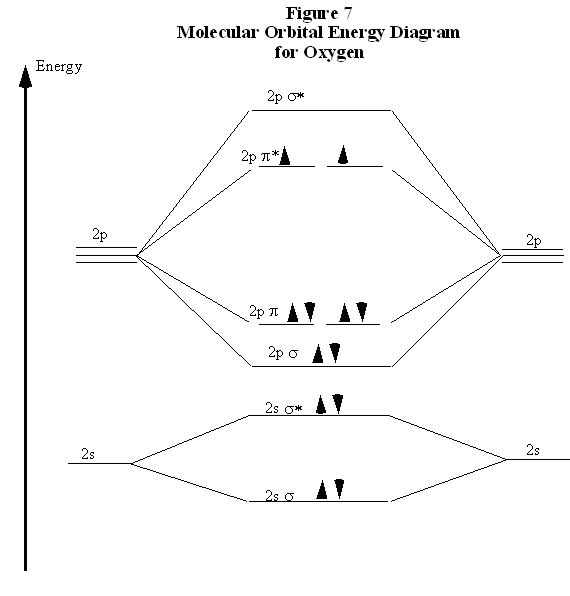
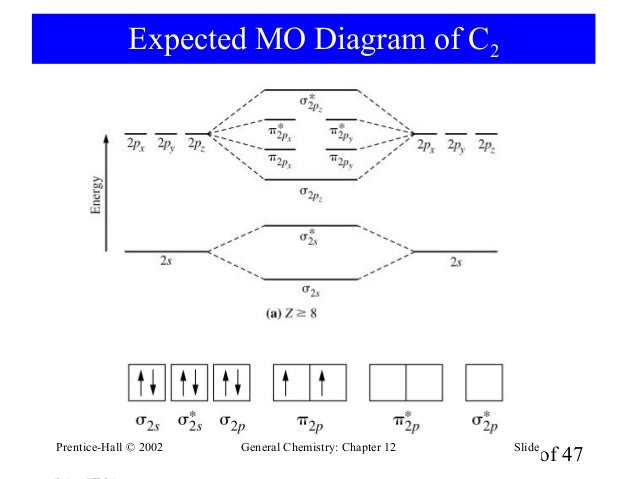
Molecular orbital diagram for carbon dimer c2. Molecular orbital diagram for n2 o2 c2 f2 also h2o. Fill from the bottom up with 8 electrons total. Mo diagrams can be used to deduce magnetic properties of a molecule and how they change with ionization. O2 2 Molecular Orbital Diagram Fabulous Electron Molecular Orbital. molecular orbitals in the diagram suggest a double bond. c. The σ2s, σ2s. *, σ2p, and σ2p ... The latter do not possess C2 rotation axes coincident to the.29 pages Molecular Orbital Theory shows that there are two sets of paired electrons in a degenerate pi bonding set of orbitals. This gives a bond order of 2, meaning ...5 answers · 8 votes: C2 exists, but only above 3,642 °C (6,588 °F) i.e. in vapor state
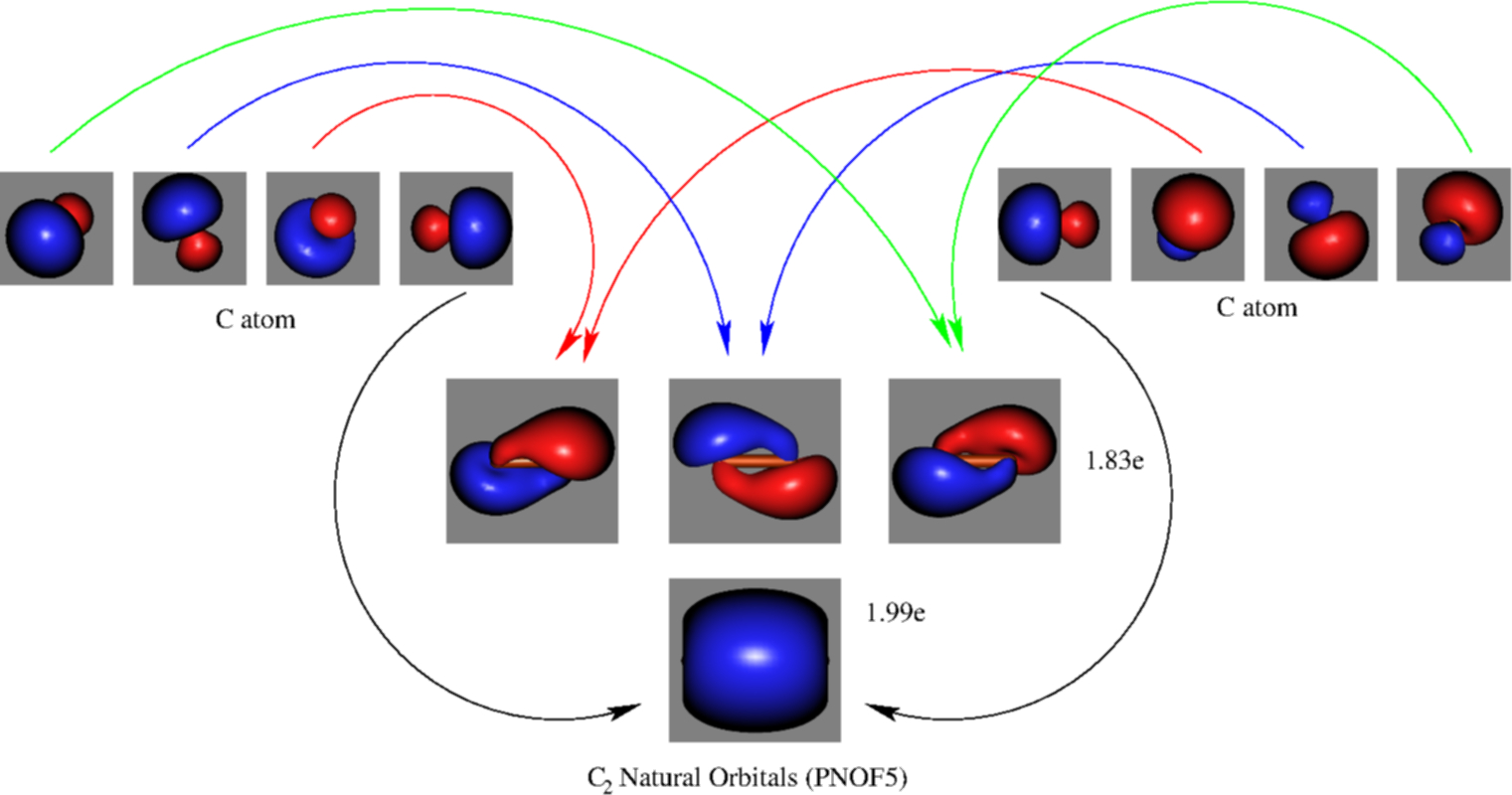
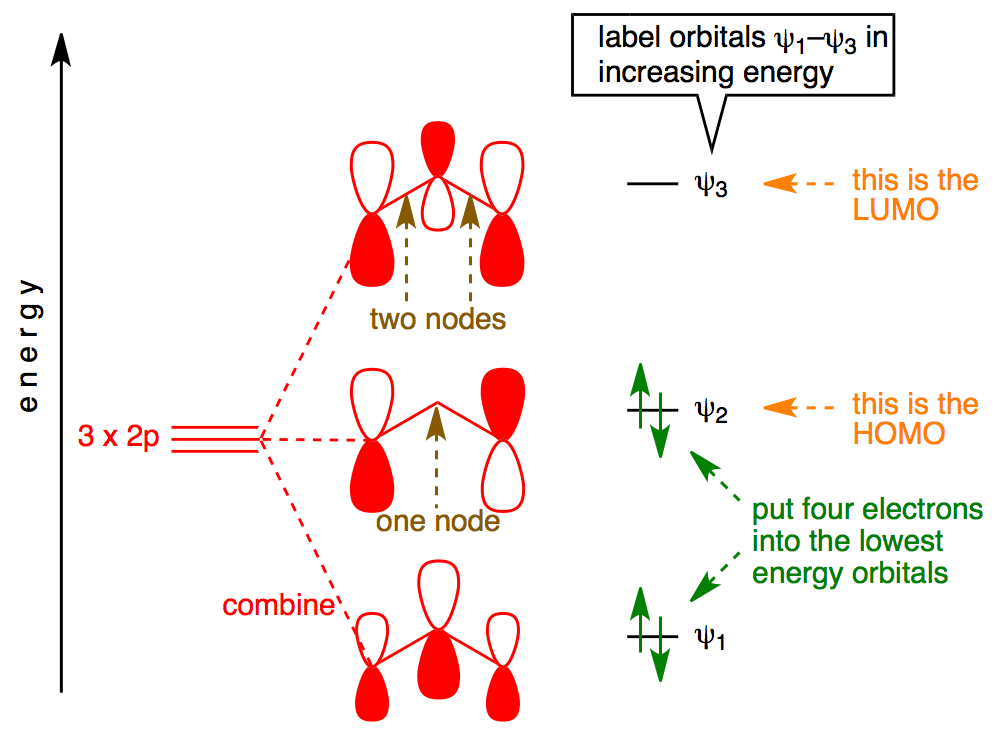
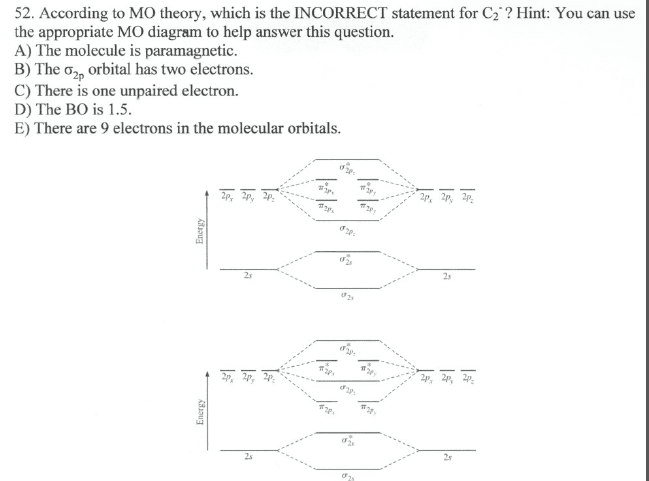
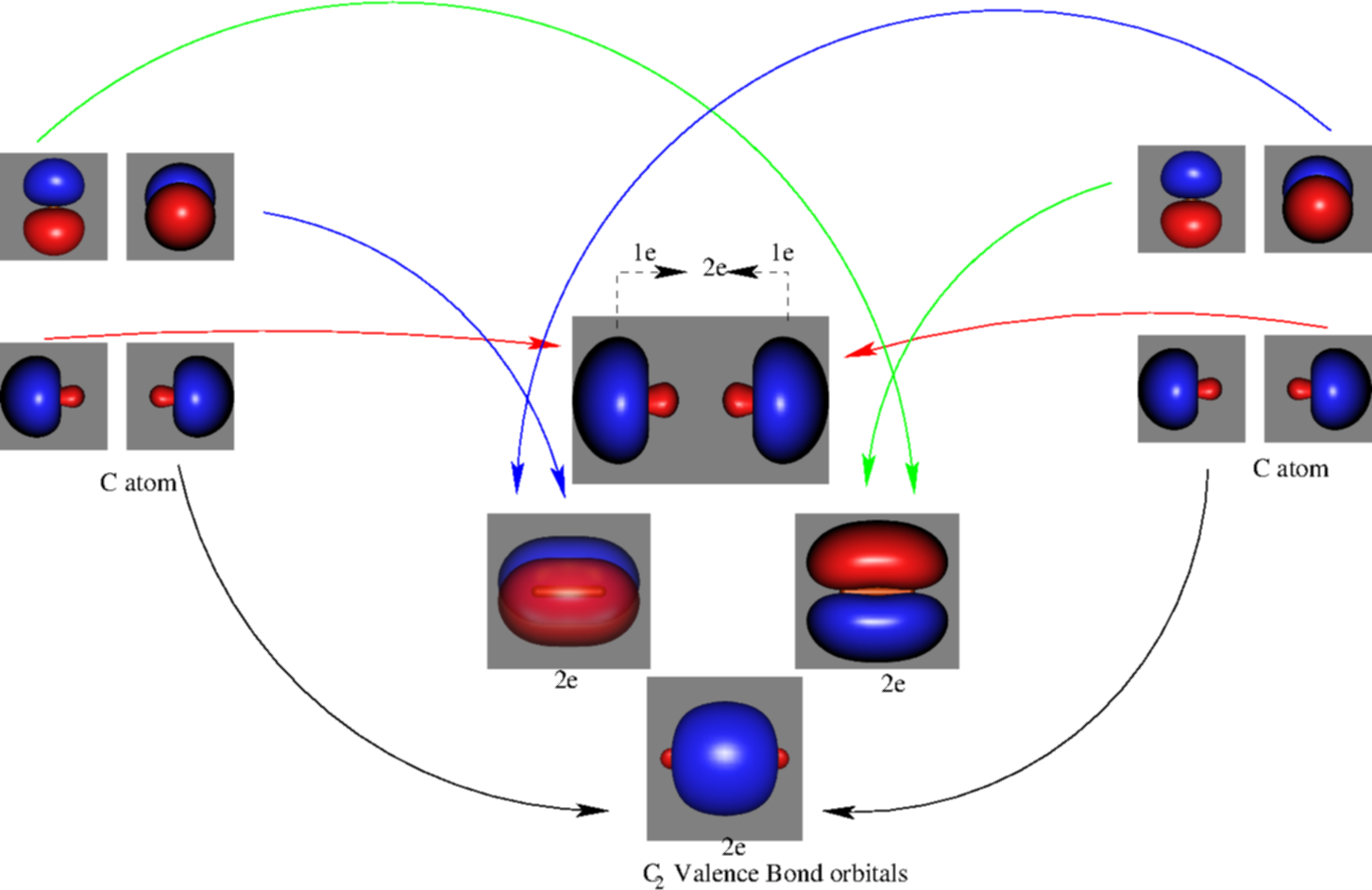
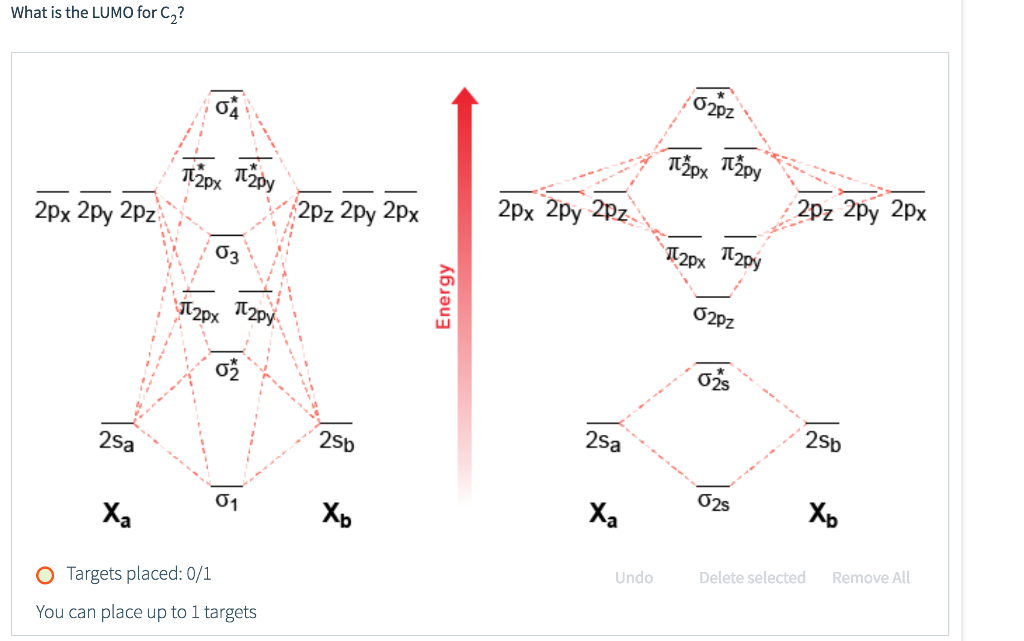
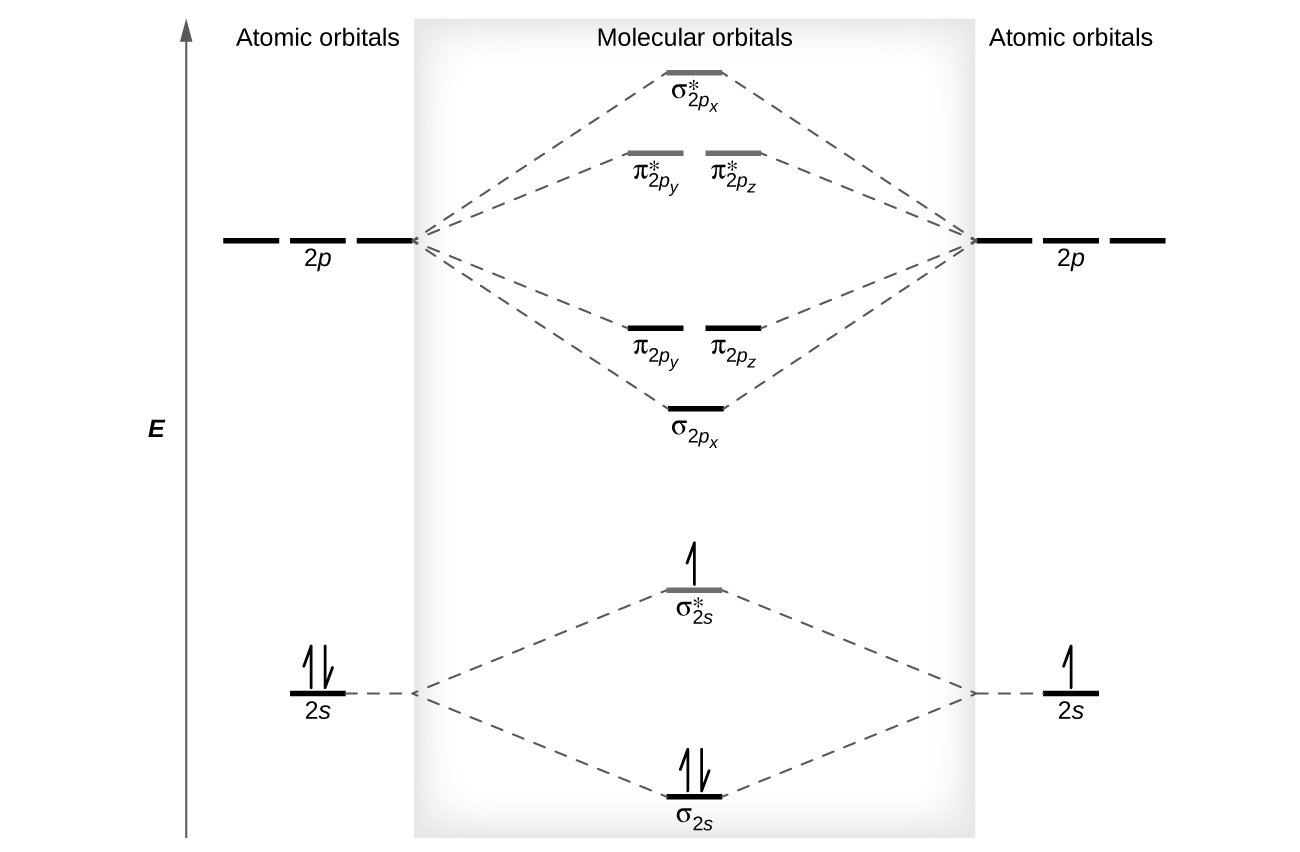
C2- molecular orbital diagram. Molecular orbital diagram for c2. This video shows the mo diagrams of the c2 n2 o2 and f2 molecules. Molecular orbitals are formed combining similar atomic orbitals. Just because some chemical species shows integral value of bond order doesnt mean that it should exist. Molecular orbital diagram for the molecule oxygen o2. C2 molecular orbital diagram. A mo is defined as the combination of atomic orbitals. As for bond orders it is 12e in bonding orbitals e in antibonding orbitals. The only orbitals that are important in our discussion of molecular orbitals are those formed when valence shell orbitals are combined. Molecular orbital theory shows that it has two sets of paired electrons in a degenerate $\pi $- bonding set of orbitals. This gives a bond order of 2, which ... The lowest energy unoccupied molecular orbital is 2pσ, so that is where the extra electron will be added. The electron configuration of the neutral C2 molecule is -- I'll use the notation given to you in the diagram. C2:(1sσ)2(1s* σ)2(2sσ)2(2s* σ)2(2pπ)4. The electron configuration of the C− 2 ion will be.
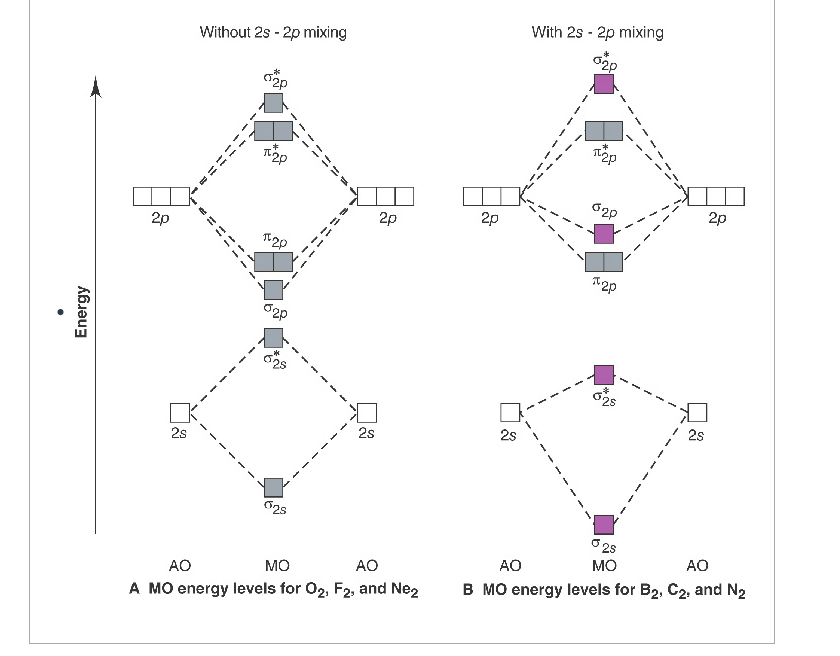
Molecular orbital diagram for n2 o2 c2 f2 also h2o. Consider the h 2 molecule for example. Sp mixing causes the σ g and σ u mos to be pushed apart in energy. Molecular orbital diagram for n2 o2 c2 f2 also h2o this problem has been solved. A diatomic molecular orbital diagram is used to understand the bonding of a diatomic molecule. The answer is C2- because of bond orders. When we draw the C2 MO, we have everything up till the PiPy Orbitlal filled, and the next orbital tht would be filled would be the sigma2Pz orbital. As for bond orders it is 1/2* [ (#e- in bonding orbitals)- (#e- in antibonding orbitals)] Doing this, normally just C2 is 1/2* [ (8)-4]=2. C2 2 molecular orbital diagram. Ion predictions with mo diagrams. Interact and form molecular orbitals. As for bond orders it is 12e in bonding orbitals e in antibonding orbitals doing this normally just c2 is 128 42. Molecular orbital diagram for carbon dimer c2. Give the molecular orbital configuration for the valence electrons in cec22. Molecular orbital diagram for c2. Molecular orbital diagram for carbon dimer c2. Get 11 help now from expert chemistry tutors. In chemistry molecular orbital mo theory is a method for determining molecular structure in which electrons are not assigned to individual bonds between atoms but are treated as moving under the influence of the nuclei ...
Molecular Orbital Theory shows that there are two sets of paired electrons in a degenerate pi bonding set of orbitals. This gives a bond order of 2, meaning ...5 answers · 8 votes: C2 exists, but only above 3,642 °C (6,588 °F) i.e. in vapor state molecular orbitals in the diagram suggest a double bond. c. The σ2s, σ2s. *, σ2p, and σ2p ... The latter do not possess C2 rotation axes coincident to the.29 pages Molecular orbital diagram for carbon dimer c2. Molecular orbital diagram for n2 o2 c2 f2 also h2o. Fill from the bottom up with 8 electrons total. Mo diagrams can be used to deduce magnetic properties of a molecule and how they change with ionization. O2 2 Molecular Orbital Diagram Fabulous Electron Molecular Orbital.

Molecular Orbital Theory Chemistry Libretexts In 2021 Chemistry Textbook Geometry Worksheets Electron Configuration

The Photoelectron Spectrum Of C2 Has Not Yet Been Measured Sketch A Predicted Spectrum Based On The Molecular Orbital Homeworklib















0 Response to "41 c2- molecular orbital diagram"
Post a Comment